It’s not a good idea to store your B12 in syringes for injections at a later date. It’s not so much about sterility, but about photolysis, which is – destruction by light. Even though vitamin B12 is extremely stable when in dry form, it is also extremely light-sensitive in liquid form, and it is easily destroyed when exposed to light.
For that reason, we sell our injectable methyl B12 in dry, powder form. This ensures the B12 hasn’t been spoiled by light or high temperatures during delivery. Photolysis is also why – after you dissolve the B12 in saline and turn it into liquid form – we recommend to wrap the vial in aluminum foil. Our vials are made of amber glass, which blocks about 66% of light. Covering with aluminum foil will completely protect the vial from light.
But, what happens when you draw the dissolved B12 into a transparent syringe each time you want to inject? Is the B12 compromised? That’s a concern we hear about quite often, such as in this email we received from Jeff, one of our customers:
“How quickly does the B12 degrade when exposed to light? I’m curious how fast I need to inject the needle once it’s drawn with fluid. I took about 2 minutes to line up and finish the first injection. Did that have any significant effect on the supplement?”
The truth is, there is no straightforward answer. But here’s some research:
Photolysis of Vitamin B12
The rate of destruction of crystalline vitamin B12 in neutral aqueous solutions, when exposed to direct sunlight, indirect sunlight, dim daylight, artificial light, ultraviolet light, and various monochromatic light sources has been determined. Sunlight, at a brightness of 8,000 foot-candles, causes a ten per cent loss for each half hour of exposure. Below 300 foot-candles, no destruction is noticeable. Artificial light of about 14,000 foot-candles causes a loss of about twelve per cent per half hour. A brightness of 3,600 foot-candles causes no alteration after two hours. With ultraviolet light the pattern of destruction is similar to that obtained with sunlight. For monochromatic light sources, photolysis is higher in the short wave length region. No destruction was observed in the red.
PHOTOLYSIS OF VITAMIN B12
Methylcobalamin is a highly photolabile and unstable molecule and hence, studies regarding photodegradation of methylcobalamin were carried out. In order to investigate the stability studies, the drug was subjected to photodegradation by exposing it to different light conditions in the validated photostability chamber as per ICH Q1B guideline. The drug was found to be less degraded in the blue light and was more prone to degradation under fluorescent light. Validated stability indicating liquid chromatography method was used for separating the methylcobalamin and its degradation products. The methylcobalamin peak with a retention time of 2.978 min was observed to decrease with a commensurate increase in a degradant peak at 4 min. The observed degradant peak was suspected to be hydroxocobalamin and was further confirmed by molecular weight determination.
Photodegradation of Methylcobalamin and Its Determination in a Commercial Formulation
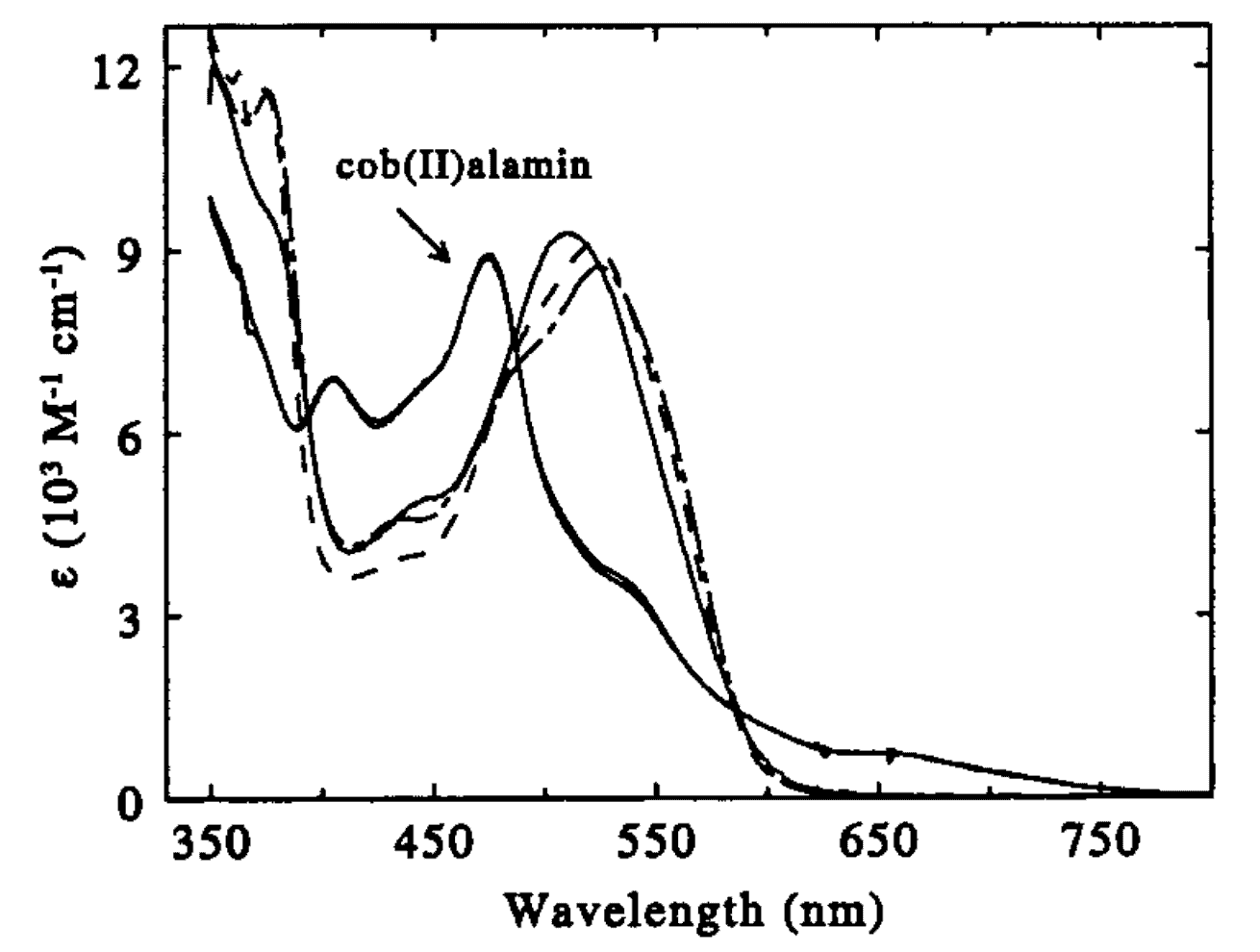
This paper presents a transient absorption study of the primary photolysis of ethyl- and n-propylcobalamin in water and a comparison of the photolysis of methyl-, ethyl-,n-propyl-, and adenosylcobalamin at two different excitation wavelengths. Excitation of methyl-, ethyl-, and n-propyl cobalamin at 520 nm in the low-lying αβ absorption band results in bond homolysis proceeding via a bound cob(III)alamin MLCT state. Theprincipal distinction between methyl-, ethyl-, andn-propyl-cobalamin lies in the relative stability of this MLCT state, with the terminal alkyl groups in ethyl and n-propyl acting as electron donors to destabilize the partial carbon anion in the MLCT state. The lifetime of the MLCT state is inversely related to the size of the alkyl ligand: 1 ns (methyl), 48 ps (ethyl), 32 ps (n-propyl), and ≤ 14 ps (adenosyl, if the MLCT state is present).Excitation at 400 nm opens a pathway for direct photohomolysis of the carbon-cobalt bond in methyl-, ethyl-, and n-propylcobalamin. This direct dissociation pathway is significant for methylcobalamin, and dominant for ethyl- and n-propylcobalamins. In contrast the photolysis of adenosylcobalamin in aqueous solution is independent of wavelength for excitation at 400 and 520 nm. The photolysis of adenosylcobalamin is also distinguished by the presence of an additional intermediate with a 110 ps lifetime that has no analogue in the other cobalamins studied.
Time-Resolved Spectroscopic Studies of B12Coenzymes: A Comparison of the Primary Photolysis Mechanism in Methyl-,Ethyl-,n-Propyl-, and 5′-Deoxyadenosylcobalamin
The photodecomposition of methylcobalamin has been studied in aqueous solution at different oxygen concentrations. In the presence of excess oxygen methylcobalamin is photolyzed at a rapid rate to yield aquocobalamin and formaldehyde as the major products. On the other hand, photolysis in the presence of only a trace of oxygen is slow and yields vitamin B12, formaldehyde, methane, and ethane. The yield of methanol is small and is not affected to an appreciable extent by the oxygen concentration. At limiting oxygen concentrations many side reactions take place which probably lead to modification of the corrin nucleus.
…
It is clear that photolysis of methylcobalamin in the presence of only a trace of oxygen is very complex. At moderate light intensities the rate of photolysis is very low, probably due to the recombination of vitamin B12 and the methyl radical. The aqueous solvent may affect the photolysis by providing a “solvent cage” around the photolyzed molecule and thus favor recombination of the radicals (Hammond and Turro, 1963). At high light intensity many side reactions occur, and more than 30% of the radioactivity cannot be accounted for. The results presented above indicate that anaerobic photolysis of the alkylcobalamins does not only yield vitamin B12 but several alkylated vitamin B12 derivatives.
The Photolysis of Methylcobalamin
Methylcobalamin is one of four members of the vitamin B12 family. We recently discovered the ability of methylcobalamin to spontaneously convert to hydroxocobalamin in aqueous solutions (Yefimov S, 2022). This discovery made it clear why the chromatograms of methylcobalamin solution show the presence of hydroxocobalamin. The light sensitivity of methylcobalamin has been known for a long time (Mehmood Y et al., 2015; Chamle A et al., 2019), so all manipulations with methylcobalamin and its solutions are carried out in a dark room. Since the process of converting methylcobalamin to hydroxocobalamin in solution is continuous, it is natural to carry out a quantitative analysis of methylcobalamin together with hydroxocobalamin. The work is devoted to the validation of this method
Analysis of Methylcobalamin and Hydroxocobalamin by HPLC/MS. Method Validation
This Perspective Article highlights recent theoretical developments, and summarizes the current understanding of the photolytic properties of cobalamins from a computational point of view. The primary focus is on two alkyl cobalamins, methylcobalamin (MeCbl) and adenosylcobalamin (AdoCbl), as well as two non-alkyl cobalamins, cyanocobalamin (CNCbl) and hydroxocobalamin (HOCbl). Photolysis of alkyl-cobalamins involves low-lying singlet excited states where photodissociation of the Co-C bond leads to formation of singlet-born alkyl/cob(II)alamin radical pairs (RPs). Potential energy surfaces (PESs) associated with the low-lying excited states as a function of both axial bonds, provides the most reliable tool for initial analysis of the photochemical and photophysical properties of cobalamins. Due to the complexity, and size limitations associated with the cobalamins, the primary method for calculating ground state properties is density functional theory (DFT), while time-dependent DFT (TD-DFT) is used for electronically excited states. For alkyl cobalamins, energy pathways on the lowest singlet surface, connecting metal-to-ligand charge transfer (MLCT) and ligand field (LF) minima, can be associated with photo-homolysis of the Co-C bond observed experimentally. Additionally, energy pathways between minima and seams associated with crossing of S1 /S0 surfaces, are the most efficient for internal conversion (IC) to the ground state. Depending on the specific cobalamin, such IC may involve simultaneous elongation of both axial bonds (CNCbl), or detachment of axial base followed by corrin ring distortion (MeCbl). The possibility of intersystem crossing, and the formation of triplet RPs is also discussed based on Landau-Zener theory.
Photolytic Properties of Cobalamins: A Theoretical Perspective
…
The photo-homolysis of the Co-C bond in alkyl cobalamins, such as MeCbl or AdoCbl, was first noted fifty years ago. Since then, the ability to generate radicals via light has become the subject of vast experimental research. It is now generally accepted that the photolysis of alkyl cobalamins results in formation of cob(II)alamin and alkyl radical pairs, similar to the intermediates from (AdoCbl)-dependent enzymatic catalysis. On the other hand non-alkyl cobalamins are generally photostable, with HOCbl being the exception, when using short wavelength excitations, which generate hydroxyl radicals. The photolysis of cobalamins depends upon a number of factors, such as the nature of both axial ligands, the local environment, and the excitation wavelengths applied. The mechanisms of photolysis at the molecular level represent a complex problem, and only recently has it become possible to use computational methods to understand the photolytic properties of cobalamins, and provide a detailed comparison with experiment. Preliminary results for the four cobalamins in this Perspective are very promising, and provide a more in-depth understanding of recent experimental results.
A mechanism of Co−C bond photolysis in the base-off form of the methylcobalamin cofactor (MeCbl) and the influence of its axial base on Co−C bond photo-dissociation has been investigated by time-dependent density functional theory (TD-DFT). At low pH, the MeCbl cofactor adopts the base-off form in which the axial nitrogenous ligand is replaced by a water molecule. Ultrafast excited-state dynamics and photolysis studies have revealed that a new channel for rapid nonradiative decay in base-off MeCbl is opened, which competes with bond dissociation. To explain these experimental findings, the corresponding potential energy surface of the S 1 state was constructed as a function of Co−C and Co−O bond distances, and the manifold of low-lying triplets was plotted as a function of Co−C bond length. In contrast to the base-on form of MeCbl in which two possible photodissociation pathways were identified on the basis of whether the Co−C bond (path A) or axial Co−N bond (path B) elongates first, only path B is active in base-off MeCbl. Specifically, path A is inactive because the energy barrier associated with direct dissociation of the methyl ligand is higher than the barrier of intersection between two different electronic states: a metal-to-ligand charge transfer state (MLCT), and a ligand field state (LF) along the Co−O coordinate of the S 1 PES. Path B initially involves displacement of the water molecule, followed by the formation of an LF-type intermediate, which possesses a very shallow energy minimum with respect to the Co−C coordinate. This LF-type intermediate on path B may result in either S 1 /S 0 internal conversion or singlet radical pair generation. In addition, intersystem crossing (ISC) resulting in generation of a triplet radical pair is also feasible.
Mechanism of Co−C Bond Photolysis in Methylcobalamin: Influence of Axial Base
Antivitamins B 12 represent an important class of vitamin B 12 analogues that have gained recent interest in several research areas. In particular, 4-ethylphenylcobalamin (EtPhCbl) and phenylethynylcobalamin (PhEtyCbl) exemplify two such antivitamins B 12 which have been characterized structurally and chemically. From a spectroscopic point of view, EtPhCbl is photolabile with a very low quantum yield of photoproducts, while PhEtyCbl is incredibly photostable. Herein, DFT and TD-DFT computations are provided to explore the photolytic properties of these compounds to shed light on the electronic properties that are indicative of these differences. Potential energy surfaces (PESs) were constructed to investigate the mechanisms of photodissociation leading to radical pair (RP) formation and the mechanisms of deactivation to the ground state. The S 1 PESs for each antimetabolite contain two energy minima, one being the metal-to-ligand charge transfer (MLCT) and another the ligand- field (LF) state. There are two possible pathways for photodissociation that can be identified for EtPhCbl but only one (path B) is energetically feasible and involves the lengthening of the Co−N Im bond through the MLCT region followed by the lengthening of the Co−C bond through the LF region. For PhEtyCbl, there is not an energetically favorable path for photolysis; rather, internal conversion (IC) is the significantly preferred photophysical event.
…
In conclusion, we have provided a theoretical analysis of the photolytic properties of two newly characterized non-natural cobalamins. These theoretical results provide insights into the electronic structure of the lowest excited state, which directly determines photolability and conversely photostability of the investigated antivitamins. There are two pathways for photolysis, and based on energetics, an optimum path can be determined. EtPhCbl (Im-[Co III (corrin)]-EtPh + ) can photodissociate via path B through the LF state with a substantially elongated axial base, although the quantum yield of photo-products is low. Similarly for PhEtyCbl (Im-[Co III (corrin)]- PhEty + ), two pathways can be identified; however, neither are energetically feasible for Co−C bond photolysis, thus explaining the incredible photostability of this antimetabolite. These results for the model complexes have important implications for understanding experimental results. As a result of the overwhelming energetic favorability for IC, PhEtyCbl is photostable. In contrast, for EtPhCbl, there is a competition of sorts between IC and photodissociation as the energetic barriers are comparable. It is not surprising that EtPhCbl and PhEtyCbl both exhibit difficulty in dissociating the upper axial ligand as these cobalamins have been shown to be metabolically inert. Photohomolysis of the Co−C bond will result in a photoactive metabolite with potential medicinal applications. In addition, the results gathered in this study are of particular importance especially as more applications for synthetic vitamin-B 12 derivatives continue to be realized.
Photolytic Properties of Antivitamins B12
So, How Long Can B12 Be In a Syringe?
As you see, it’s hard to say precisely, because it depends on the form of B12 that you’re using, the wavelength of light, and other factors. But, what we do know from our own personal experience with thousands of customers, is that normal usage (drawing from an aluminum-wrapped vial, and injecting within a minute or two) does not render the B12 even remotely ineffective. Far from it. Our customers regularly expose their B12 to light for a couple of minutes each time, and still experience tremendous improvements.
Just keep these suggestions in mind:
- If your B12 vial came pre-dissolved (unlike when you buy from us), then immediately cover it with aluminum foil to block light away.
- Remember, it’s better to buy B12 in dry form, because in this way it’s remarkably stable to both heat and light. This will protect the vitamin during delivery.
- When you give yourself vitamin B12 injections at home, store the vial in the fridge or any cool environment of 5-25°C / 41-77°F degrees.
That’s about it.